About
Key Milestones
Inventvacc was founded by Dr. Kapre & Mr. Pradip Ghate in 2016 to develop vaccines for rare diseases currently affecting more than 400 million people worldwide.
Our mission and focus is to empower healthy living by developing unique, efficacious and affordable vaccines for the populations most in need. Over the years, the company has grown from a small R&D lab, operating out of Vancouver, Canada, to a multinational commercial operations with an innovative pipeline.
Timeline 2016 - 2026
Click on the dots below each date to navigate the timeline
Use the arrows to see more of the timeline
Team
A team of industry veterans & partners with more than 35 years’
experience in the vaccine business responsible for:

40
Vaccine
Manufacturing Plants

3,50 Billion
Doses of Vaccines
Manufactured

10 Billion
Vaccines Sold

Dr. Subhash Kapre PHD
Chairman
Subhash Kapre, PhD heads the InventVacc board as Chairman. His experience encompasses multiple senior executive vice president positions at Serum Institute of India Pvt. Ltd. (SIIPL), which measured by volume, is the world’s largest vaccine manufacturer.
During his four-decade tenure at SIIPL, Subhash held multiple functional leadership positions in R&D, manufacturing, and vaccine discovery. He is a thought leader in rDNA technology, an internationally renowned expert in both bacterial and viral vaccines and has led certification of WHO- Prequalification (regulatory approval) for large scale manufacturing.
He held board positions at Akorn, and Serum Institute of India, where he was board, executive director. Subhash has a history of driving multiple vaccines from discovery, into development and through licensure for infectious diseases, including MenAfriVac® and Pneumosil®, conjugate vaccines which together will represent over 500M aggregate doses delivered by 2022. Subhash holds over 70 patents and founded Inventprise in 2012 to continue his career long mission to provide vaccines for those most in need.

Pradip Ghate
Chief Operating Officer
Pradip Ghate has over three decades of international experience working in developing and developed countries in Project and Operations management for Aseptic Sterile operations of biological and pharmaceutical products.
He has direct experience managing multiple disciplines from Aseptic manufacturing, Quality Assurance, Quality Engineering and Systems Validation, Formulation, Fill, Finish & Packaging.
His tenure at Serum Institute of India, Hemosol Inc, Cardinal Health, Catalent, and now Inventprise LLC consists of progressively increasing responsible titles ranging from Manufacturing head, project management to Chief Operating Officer.
At InventVacc, Pradip is overseeing the day to day administrative and operational functions of our business. Pradip is a Certified Lean Six Sigma Black Belt and believes in Operational Excellence. His strategy planning and vision is the motive force behind operational objectives at InventVacc.

Luis Baretto M.B.B.S., M.D., M.H.Sc
Senior Scientific Advisor
Dr. Luis Barreto is the President of Dr. Luis Barreto & Associates, a consultancy firm specializing in vaccine development, immunization, science policy, global health and biotechnology. He is a member of the Scientific Advisory Board for the Human Health Therapeutics Group of the , National Research Council (NRC), Canada, a Senior Scientific Advisor to Inventprise, U.S., a member of the Scientific Advisory Board of Cansino Biologics, China, Chair of the International Data Safety Monitoring Committee for Medicago Covid Vaccine trials, Dr Barreto is a member of the International Advisory Board of the McMaster/Maastricht/Manipal Global Health Program
Dr. Barreto was formerly Vice-President of Clinical, Medical and Regulatory Affairs, Vice-President of Immunization and Science Policy, and a member of the Senior Management Team at Sanofi Pasteur Canada, as well as a member of its Global Public Policy group. He spent more than 23 years in research and development, leading clinical trials, licensing and launching vaccines, working closely with Canadian and international investigators and policymakers, and helping to shape immunization policy in Canada and globally working closely with GAVI and WHO. Prior to joining Sanofi Pasteur Canada, he was the first Territorial Epidemiologist of the Northwest Territories.
Dr. Barreto has been/is on many volunteer boards of directors, including VIDO-InterVAC (Chair), Research Canada, March of Dimes. He has been/is on many advisory and review panels with the government of Canada and Not for Profit organisations.
Dr. Barreto is a recipient of the BIOTECanada Community Service Award, the Research Canada Award of Distinction, the BioSciences Education Canada Meritorious Service Award, and the March of Dimes Canada Paul Martin Sr. Award. He has also a recipient of the Community Service Life Sciences Ontario award.

Dr. Subhash Kapre PHD
Chairman
Subhash Kapre, PhD heads the InventVacc board as Chairman. His experience encompasses multiple senior executive vice president positions at Serum Institute of India Pvt. Ltd. (SIIPL), which measured by volume, is the world’s largest vaccine manufacturer.

Pradip Ghate
Chief Executive Officer
Pradip Ghate has over three decades of international experience working in developing and developed countries in Project and Operations management for Aseptic Sterile operations of biological and pharmaceutical products.

Luis Baretto M.B.B.S., M.D., M.H.Sc
Senior Scientific Advisor
Dr. Luis Barreto is the President of Dr. Luis Barreto & Associates, a consultancy firm specializing in vaccine development, immunization, science policy, global health and biotechnology.
Commitment to Canada
InventVacc was born In Canada and is committed to develop and manufacture Haemophilus Influenzae Type A (Hia) vaccine in collaboration with the National Research Council of Canada (NRC) and the Public Health Agency of Canada (PHAC). InventVacc is also committed to develop & manufacture innovative,
affordable vaccines in Montreal

Haemophilus influenzae type a (Hia)
Each year in Canada, especially in the North and in Indigenous communities, many infants and immunocompromised adults are at risk of Hia. The bacterial infection can result in pneumonia, meningitis, septic arthritis, and bloodstream infections.
Since the late 1990s, there has been an emergence of Hia infections, especially in Indigenous communities in the northern regions of Canada and Alaska associated with significant morbidity and approximately a 10% mortality rate.
Several cases per year are being recorded in Canada and the disease is spreading to other regions of the globe which led to the World Health Organization exploring with InventVacc and NRC to make this vaccine available.
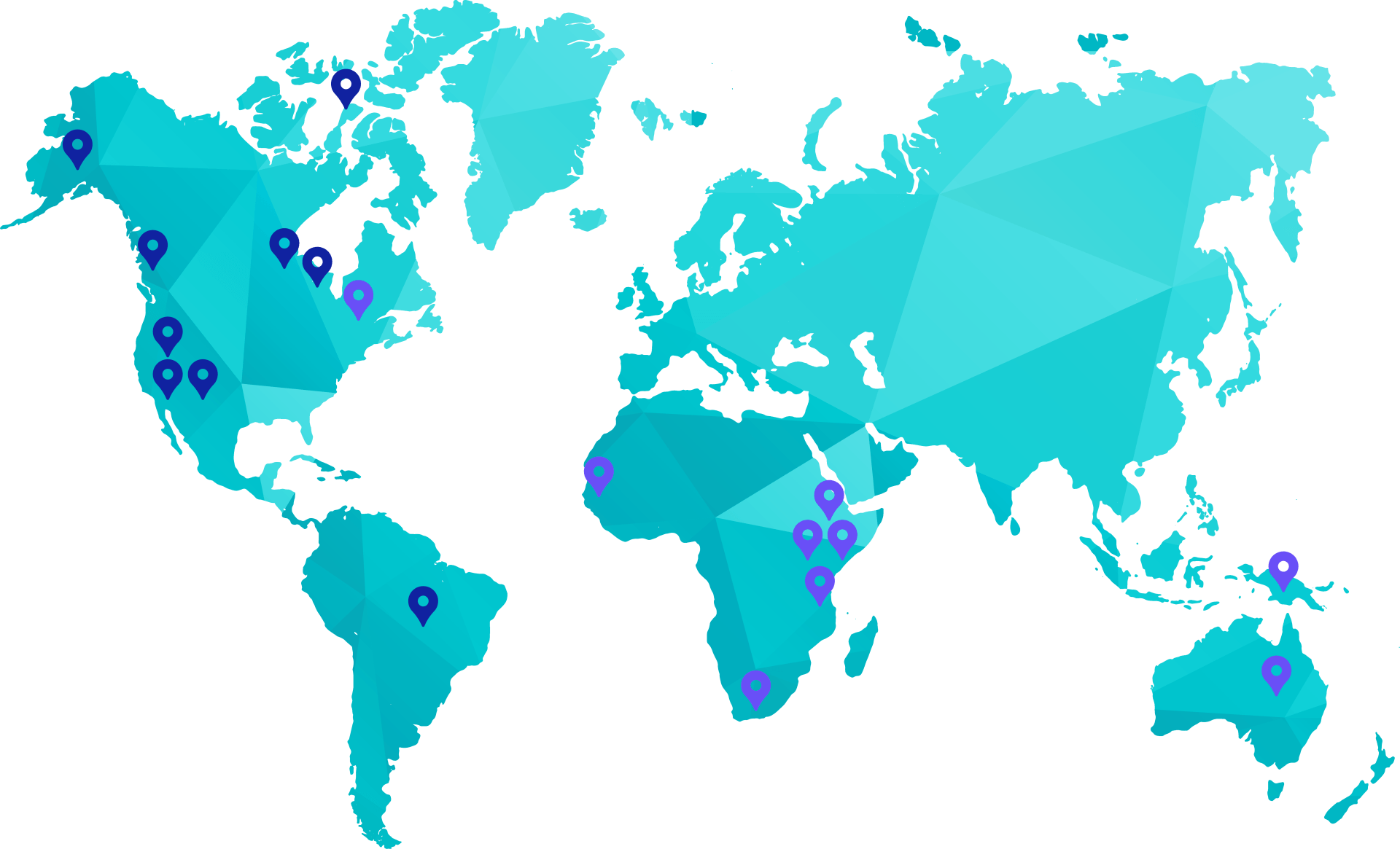
GLOBAL EPIDEMIOLOGY OF HIA
- Many reported cases
- One or few reported cases
InventVacc Biologics Inc. and its partner Inventprise are committed to developing and manufacturing vaccines for this rare and neglected disease with the objective of preventing infections and saving lives.
